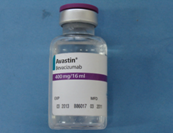
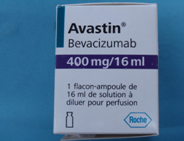
FDA Issues warning on counterfeit Avastin
The FDA is warning healthcare professionals and patients about a counterfeit version of Avastin 400mg/16mL, which may have been purchased and used by some medical practices in the United States. The counterfeit version is labeled as Avastin, manufactured by Roche and does not contain the medicine’s active ingredient, bevacizumab, which my have resulted in patients not receiving needed therapy. 19 medical practices in the United States purchased unapproved cancer medicines from Quality Specialty Products (QSP), a foreign supplier that may also be known as Montana Health Care Solutions. Volunteer Distribution in Gainesboro, Tennessee is a distributor of QSP’s products.
Pictures of the counterfeit version of Avastin are shown below:
Avastin is an injectable medicine used to treat cancer and is administered to patients in clinics, hospitals, and doctors’ offices. Roche is the company that manufactures Avastin approved for marketing outside of the United States. Roche conducted laboratory tests that confirmed the counterfeit version of Avastin. Packages or vials may be counterfeit if they:
- are labeled with Roche as the manufacturer
- display batch numbers that start with B6010, B6011 or B86017
Medical practices that have obtained products from Volunteer Distribution and QSP should stop using them and contact the FDA. These products should be retained and securely stored. To report suspect counterfeit products and other suspect products obtained from Volunteer Distribution or QSP/Montana Health Care Solutions:
- Call FDA’s Office of Criminal Investigations (OCI) at 800-551-3989,
- Visit OCI’s Web site (www.accessdata.fda.gov/scripts/email/oc/oci/contact.cfm), or
- Email – DrugSupplyChainIntegrity@fda.hhs.gov
Healthcare professionals and patients are encouraged to report adverse events related to the use of suspect injectable cancer medications to the FDA’s MedWatch Safety Information and Adverse Event Reporting Program:
- Complete and submit the report Online: www.fda.gov/MedWatch/report.htm
- Download form or call 1-800-332-1088 to request a reporting form, then complete and return to the address on the pre-addressed form, or submit by fax to 1-800-FDA-0178
Read the MedWatch safety alert, including links to the FDA Counterfeit Statement, letters to medical practices, and the company Press Release, at:
http://www.fda.gov/Safety/MedWatch/SafetyInformation/SafetyAlertsforHumanMedicalProducts/ucm291968.htm